Identify and protect yourself from sulfonic acid fraud
Sulfonic acid is one of the most widely used raw materials in the cleaning products industry due to its good cleaning performance and high foaming capacity at a relatively low cost, making it a competitive product in the market. Despite this, caution is necessary, as there are increasing reports of attempted fraud in the chemical industry. In this article, you will understand what types of fraud involve sulfonic acid, how to identify them, and how to avoid them.

Sulfonic acid:
chemical structure and applications
Sulfonic acids are a class of organic compounds formed by an alkyl carbon chain linked to a sulfonate group – (SO32-). They are obtained through a substitution reaction called sulfonation, where a hydrocarbon reacts with sulfuric acid (H2SO4) or sulfur trioxide (SO3) or chlorosulfuric acid (ClSO3H), causing the substitution of hydrogens in the carbon chain by sulfonate groups.
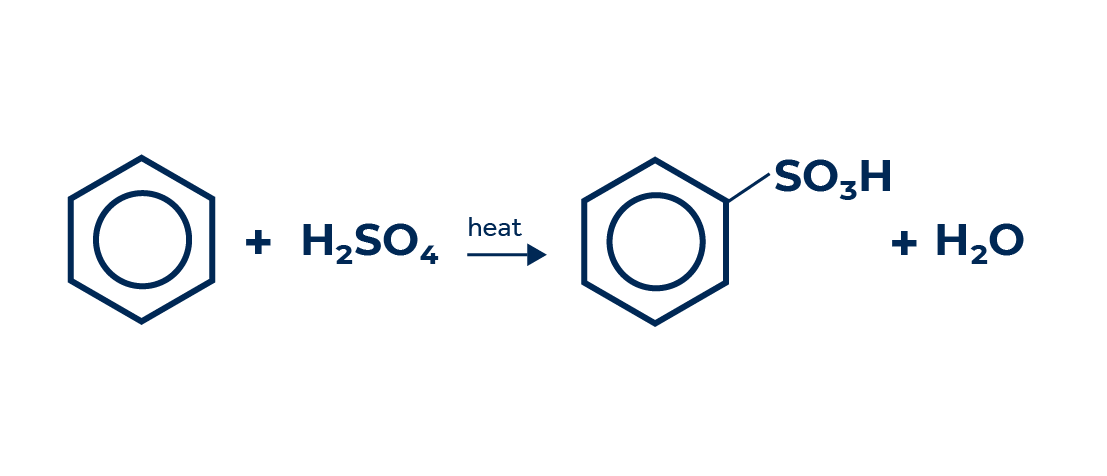
Simplified sulfonation reaction of benzene
However, the term sulfonic acid is commonly used in the chemical industry to name linear alkylbenzene sulfonic acids, typically with an alkyl chain containing 10 to 14 carbons. These are extensive molecules containing a nonpolar chain, commonly called the tail, which has an affinity for oils and fats, and another polarized region, called the head, which has an affinity for water.
This is an anionic surfactant, meaning that its polar group has a negative charge. Surfactants belonging to the anionic class exhibit high detergency, good water solubility, significant foam formation, and good cleaning performance.
Among these, sulfonic acid stands out, being one of the main raw materials used in the cleaning products industry in a wide variety of formulations, such as various detergents, general cleaners, degreasers, and multi-purpose cleaners.
Common frauds in the sale of Sulfonic Acid
The most common forms of commercialization of this input are in 90% and 96% versions, which indicate that they contain, respectively, ninety percent and ninety-six percent of active matter.
Taking 90% Sulfonic Acid as an example, this means that for every 100 kilograms, 90 kilograms are the acid itself, and the other 10 kilograms constitute the sum of byproducts of the reaction involved in the production process, impurities, and water.
However, there may be discrepancies between the declared percentage and the actual content, constituting fraud. Fraud occurs to reduce production costs and increase profit, since the sale is made according to the value of a concentrated raw material, but with a real concentration lower than that declared by the supplier.
Quality control must be efficient and rigorous in order to identify attempts at fraud, anticipating undesirable situations that may occur if a fraudulent batch is used as raw material in the process. However, it is necessary to better understand how these attempts to adulterate the product occur and what methods are used in this operation.
Generally, water is added to sulfonic acid, diluting it to concentrations lower than those indicated. It is known that for an addition of up to 10% water, the product does not undergo major changes in its physical characteristics and appearance. However, when adding quantities greater than 10%, its rheology is altered, and what was previously a slightly viscous liquid takes on an appearance closer to that of a gel.
Then, sulfuric acid is used in the dilution process, which allows for the addition of slightly larger quantities than ten percent of water, further diluting the concentration of the active ingredient in the product.
How to identify fraud?
The following are the mechanisms and analyses that can be used to detect the altered input by modifying its concentration.
Analysis of the active ingredient content or active substance content:
Since it is an anionic surfactant, it is possible to measure the active ingredient content using a two-phase titration method. To learn more about this methodology and download the step-by-step analysis guide, click here.
Determination of free sulfuric acid:
It is important to quantify the free sulfuric acid, which, in addition to being a byproduct of the sulfonic acid production reaction, can also be added along with water to further dilute the active ingredient content, as previously explained.
The method employed in this analysis consists of a titration, in the presence of Dithizone indicator, with a lead nitrate solution of known concentration that will react with the sulfate ions present in the reaction medium. When these ions are completely consumed, the addition of excess lead nitrate will cause the lead to react with the indicator, forming a plumbic complex, revealing the red color that indicates the titration endpoint. Through the volume of lead nitrate solution consumed, the content of free sulfuric acid is determined by stoichiometric calculations.
To learn the step-by-step process for determining free sulfuric acid, download the methodology by clicking on the image below:
Learn how Macler prevents
fraud with TrustScan
Macler is known for paying extreme attention to the quality of its products. We do this through TrustScan®, an internal procedure that reinforces our commitment to delivering performance-guaranteed products to customers and partners. We go beyond traditional physicochemical analyses.
Keep reading to learn more.
Application tests:
To ensure efficiency during production, application tests are carried out on standard formulas suitable for each raw material, analyzing their behavior in practice and avoiding unexpected problems for the process or product.
In this test, the sample is subjected to laboratory-scale manufacturing of a product in which the raw material is used; that is, it is applied in a real-world situation, but on a reduced scale. In the case of sulfonic acid, it could be applied in a detergent formulation, for example.
Variations in the concentration of the active ingredient influence the solubility of the medium and consequently affect the physicochemical properties of the formulation, which can be detected through simple analyses, such as measuring viscosity.
The higher the concentration of the active ingredient, the greater the amount of solute present, making the medium less soluble. This, in turn, will contribute to an increase in viscosity based on the same percentage of some electrolytic agent, usually sodium chloride or magnesium sulfate, salts widely used as thickeners.
Carbon chain dispersion analysis:
An analysis that reveals the quantities present of each specific molecule and is performed through chromatography coupled to a mass spectrometer. These are robust, precise, and technologically advanced pieces of equipment, and therefore, the investment in their acquisition, operation, and maintenance is high and only justifiable if the volume of analyses is very high.
At Macler, a partnership with a third-party laboratory was developed to perform carbon chain dispersion analysis on a sample of 90% sulfonic acid. The reason for this investigation was that, during the analysis of the active matter content, a value within the specification range was found, but in the application test, it was not possible to achieve the desired viscosity, indicating that there was some problem with the batch.
Thus, the importance of TrustScan® becomes clear in practice, revealing that this sample, when used in any of its applications, would not behave in a known manner, and consequently, the properties of the product formulated from it could also be impacted.
Finally, the dispersion analysis revealed the unusual behavior of this batch. The percentage of the actual active ingredient was well below the specified amount, while a higher quantity of impurities was present. These impurities have shorter carbon chains compared to the sulfonic acid chain and lower molecular weight; therefore, the molecular weight reported by the manufacturer was higher than the actual molecular weight, which interfered with the analysis of the active ingredient content, reporting a higher value than the real value.
Optimize your processes by using blends.
It is crucial not only to identify but also to take steps to prevent attempted scams in the market. At Macler, blend development technology emerges as a viable, safe, and efficient alternative.
Replacing commodities with blends adds value and can contribute several advantages to the process and the product. A blend can replace various raw materials within a formulation, bringing benefits such as: reduced inventory volume and savings in physical space; direct reduction in formulation costs in certain situations; and most importantly: ensuring the appropriate concentration of active ingredient for the product, preserving its characteristics, and guaranteeing the quality of manufactured batches.
.jpg)
Count on Macler to guarantee the performance of your formulations.
Ensuring the accuracy of the stated concentration is the minimum a supplier can do. Macler goes a step further. With Trustscan®, you practically guarantee the performance of your chemical solutions.
Contact our SmartLab experts to learn more.
Learn More
Our chemistry
We use our labs to create
intelligent chemical solutions balanced with your reality.
Products
We use our R&D center, our own laboratory with experienced professionals, to deliver intelligent chemical solutions in balance with your reality.


